A Hidden Inheritance in American Landfills
There is a story we tell ourselves about American industrial history that goes something like this: we used to make things out of metal, and now we make them out of plastic. The story is true as far as it goes. What it leaves out is the question of where all that metal went.
The answer, for an enormous percentage of it, is the same place: the ground.
The Forgotten Material Reality of the Mid-Century American Pantry
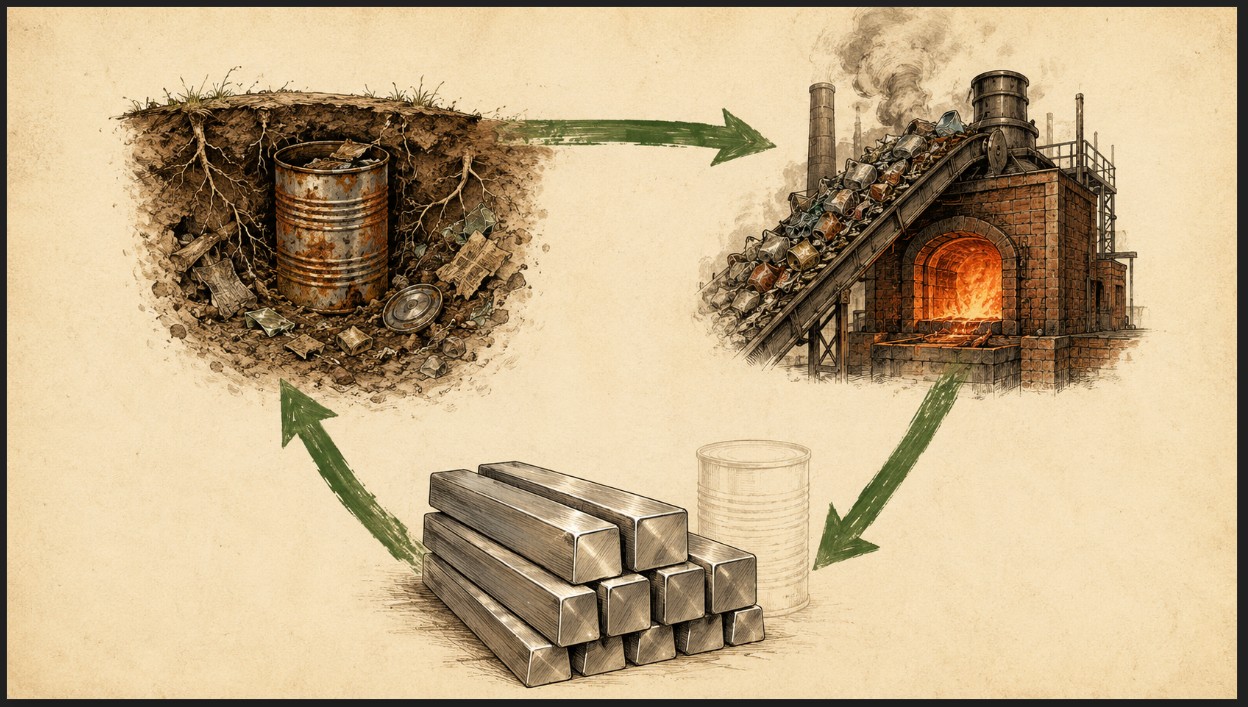
For roughly the first three quarters of the twentieth century, the standard American food container was a steel can. Not “tin can” in the loose modern sense — steel, plated with a thin layer of tin to prevent corrosion of the food inside, but structurally, mechanically, and metallurgically a steel container. The tin layer was measured in microns. The can itself was a real piece of metal, drawn or soldered from sheet steel produced by mills in places like Pittsburgh, Bethlehem, Gary, Youngstown, and — yes — Johnstown.
Aluminum did not begin to displace steel for beverage cans until the late 1950s, and did not become dominant for beverages until the 1970s. For food cans — soup, vegetables, fruit, meat, fish, condensed milk, coffee — steel held the market well into the 1980s, and it remains the standard for many food categories today. For most of the American twentieth century, the material substrate of the household pantry was high-quality, mill-rolled, food-grade carbon steel.
How much steel?
Consider the math at household scale. A mid-century American family of four, eating from a pantry stocked typically for the era, would have generated something on the order of three to seven empty steel cans per week. Call it five, conservatively. That’s roughly 260 cans per household per year. The empty weight of a standard mid-century food can was roughly 50 to 80 grams of steel, depending on size — call it 60 grams as an average. That’s 15.6 kilograms — roughly 34 pounds — of high-quality food-grade steel per household per year, going into the trash.
Now scale that up. The United States in 1950 had roughly 43 million households. By 1970, around 63 million. Across just those two decades, at the conservative household estimate above, that is somewhere between 15 and 20 million metric tons of food-grade steel discarded as empty cans into American landfills, dumps, and burn barrels.
That figure does not include:
- Coffee cans, lard cans, and bulk-format steel containers (much heavier per unit)
- Steel beverage cans before aluminum displaced them
- Industrial and commercial food packaging
- Steel paint cans, oil cans, motor product cans
- Aerosol cans (steel-bodied through most of this period)
- Cookie tins, candy tins, decorative tins (often heavier-gauge steel)
- The countless other steel containers — biscuit tins, tobacco tins, polish tins — that were the material reality of mid-century consumer life
A defensible estimate of total steel discarded through consumer packaging alone, across the twentieth century, in American landfills, lands somewhere between 100 and 200 million metric tons. That is not a typo. That is a quantity of steel comparable to the entire annual production of major steel-producing nations.
It is sitting in the ground.
What That Steel Actually Is
This is where the conventional intuition fails. The reflex assumption is that century-old steel cans in a landfill are just rust — degraded, dispersed, valueless. The chemistry says otherwise.
Steel cans rust, certainly. Iron exposed to moisture and oxygen forms iron oxides — Fe₂O₃, Fe₃O₄, FeO(OH), and related compounds. But the iron atoms are still there. They have not gone anywhere. They have changed chemical state, not chemical identity.
And here is the critical fact that gets lost: iron ore is iron oxide. The hematite, magnetite, and goethite that the world’s iron mining industry has been digging up for three thousand years are the same family of compounds as the rust on a corroded steel can. The mining industry pulls iron oxide out of rock and reduces it back to iron through smelting. That process — well-understood, mature, industrially routine — works on rust as well as it works on natural ore. Better, in many cases, because the rust on a landfilled can is often surrounded by unrusted metal beneath it, while natural iron ore is mixed with gangue minerals that have to be removed first.
A landfill stratum from 1955 is, in chemical and mineralogical terms, an iron deposit. It happens to be one that was concentrated and emplaced by industrial civilization rather than by geological processes, but the recoverable material is the same material, in the same compounds, at concentrations that compare favorably with primary mining operations.
The phrase that matters: the steel cans in the ground are iron ore that has been mined for us by time.
What This Means for Reclamation Operations
For the landfill reclamation thesis developed elsewhere in this series, this is the line item that anchors the economics. Metals recovery is the most mature of the three production streams in a reclamation operation, and within metals recovery, ferrous recovery is the most mature subdiscipline. Magnetic separation pulls iron and steel out of mixed waste with high efficiency at low cost. The recovered material — even fully oxidized — feeds existing smelter and steel mill infrastructure with no special handling beyond what these mills already do for low-grade ore and scrap blends.
The question for an operation looking at a 1950-1980 era American landfill is not “is there steel in there.” There is. Tens of thousands of tons of it per landfill, often hundreds of thousands at the larger sites. The question is how much of it is recoverable per excavated ton, and at what energy cost.
Both answers are favorable. Mid-century landfill steel is typically:
- High-grade. Food cans were made from cleaner steel than most industrial scrap, because food contact regulations required it. The base alloy was generally low-carbon steel with controlled impurities, well-suited to remelting.
- Concentrated. Cans cluster. Households dumped them by the bag. Municipal waste streams of that era were heavy with metal in ways modern waste streams are not. A mid-century stratum can run several percent ferrous content by weight — comparable to or better than a low-grade iron ore deposit.
- Accessible. Steel does not migrate the way fine particulates can. The cans are still where they were buried, often with their general structure intact even after decades of oxidation. Magnetic separation finds them readily.
The Civic Implication for Steel-Heritage Cities
For a city like Johnstown — or Bethlehem, or Pittsburgh, or Youngstown, or Gary, or any of the dozens of American steel towns that built the country and were then abandoned by it — there is a particular dignity in this argument that should not be lost.
The steel that built mid-century America was rolled in these cities. It was shipped out, made into cans, filled with food, sold across the country, eaten, and tossed. The steel that fed a generation of American households came from us. Now it sits in the ground, waiting.
A regional materials recovery operation centered on steel-heritage cities would not be reactivating those cities to do new work. It would be reactivating them to finish the work they started — to bring the steel home, melt it back down, and return it to use. The same mills, the same workforce, the same rail corridors, the same metallurgical traditions. The same material, traveling in a circle that took seventy years to close.
This is not nostalgia. It is supply chain economics. The steel exists. The infrastructure to process it exists. The skilled labor exists. The proximity of the steel to the processing capacity exists. The market for recycled steel exists and is growing. Every link in the chain is in place except the political and capital decision to close the loop.
What “Reuse” Actually Looks Like at This Scale
It is worth being concrete about what 100 to 200 million metric tons of recoverable steel from twentieth-century American landfills represents in terms of useful work.
That quantity of steel, refined and rolled, is enough to:
- Build the structural steel for several hundred thousand mid-rise buildings.
- Manufacture tens of millions of automobiles.
- Produce the rails for several complete national rail networks.
- Provide the reinforcement steel for a generation of infrastructure construction.
These are not all things that need to happen, and they are not all things that should happen all at once. But they describe the scale of the latent material wealth in question. It is not a niche. It is not a marginal supplement to primary steel production. It is, potentially, a substantial fraction of American steel demand for decades, sourced from material that has already been paid for, already been refined once, and is currently sitting in the ground producing nothing but methane and leachate.
The American twentieth century built itself in steel and threw most of the steel away. We are now in a position to recover it.
The Frame
The story we tell ourselves about industrial history — that the metal age ended and the plastic age began — is true at the surface and false underneath. The metal did not stop existing. It stopped being visible. It went into the ground.
It is still there. It has not gone anywhere. The chemistry says so, the mining-industry analogy confirms it, the basic conservation of mass guarantees it, and the technology to recover it has been industrial standard for longer than any of us has been alive.
The tin can was steel. The steel is still there.
We just have to go get it.
Observe. Design. Intervene.